Research Article
Evaluation of Modifiable Factors Leading to Intraoperative Contamination from Surgical Helmet Systems
David B. Johnson1*, Tyler Noble2, Jacob J. Triplet1 and David Maish2
1Doctors Hospital OhioHealth, 5100 West Broad Street, Columbus, OH 43228
2Wellspan York Hospital, 1001 S. George St York, PA 17403
*Address for Correspondence: David B. Johnson, Doctors Hospital OhioHealth, 5100 West Broad Street, Columbus, OH 43228. Tel: +407-221-5236; E-mail: David.Johnson4@ohiohealth.com
Dates: Submitted: 10 August 2018; Approved: 22 September 2018 2018; Published: 25 September 2018
Citation this article: Johnson DB, Noble T, Triplet JJ, Maish D. Evaluation of Modifiable Factors Leading to Intraoperative Contamination from Surgical Helmet Systems. Int J Ortho Res Ther. 2018;2(1): 008-012.
Copyright: © 2018 Johnson DB, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
Background: Periprosthetic Joint Infections (PJI) pose significant challenges to the patient, surgeon and health care system. With a continued rise in arthroplasty procedures, methods to minimize PJI should be explored. Recent literature has demonstrated increased contamination at the Gown Glove Interface (GGI) using modern surgical toga systems, predisposing to PJI. The role of modifiable risk factors for contamination such as helmet fan speed, toga waist tightness, and gloving techniques are unknown. Here, we evaluate fluorescent particle egress at the GGI following modification of sterile gowning technique for surgical helmet systems.
Methods: Twelve standardized Total Knee Arthroplasty (TKA) simulations were performed utilizing Total Shield Surgical Togas and helmets. Fluorescent powder and ultraviolet light were used as a means of detecting contamination prior to, and following, standard double-glove gowning and compared with modifications in helmet fan speed, toga waist tightness, and a commonly used alternate gloving technique.
Results: No particle contamination was identified outside of the outer-glove for any simulation irrespective of fan speed or toga waist tightness. However, powder was noted to have migrated through the pervious elastic wrist cuff and onto the gown nearing the sterile boundary when a standard double gloving technique was utilized. The commonly used alternate gloving technique, of under-gloves placed prior to the toga, revealed no particle egress through the cuff or near the GGI.
Conclusion: Alternate gloving may minimize contaminant egress. Fan speed and toga waist tightness have little effect on particle egress at the GGI.
Introduction
Periprosthetic Joint Infections (PJI) pose a significant burden to the patient and surgeon, while adding a substantial cost to the health care system [1]. Paralleling the aging population, the total number of joint arthroplasties completed annually continues to rise; and with it, the total number of periprosthetic joint infections [2]. It is estimated that by 2030, 3.48 million total knee arthroplasties will be performed annually in the United States; a 673% increase from 2005 [3]. An increase in the number of PJI will likely have a significant economic impact, an estimated $1.62 billion annually by 2020 [1]. Moreover, PJI are the most common cause of failure in both primary and revision Total Knee (TKA) and Total Hip Arthroplasties (THA) [4,5], with early series reporting infection rates as high as 9.5% [6]. Wound contamination at the time of the operation is believed to be the primary source in most early postoperative infections [7-11]. Shockingly, previous reports have demonstrated that bacterial laden cells shed from operating room staff are thought to be the primary source of contamination in 98% of cases [5,9,10]. Thus, modifiable factors that may minimize contamination from operating room personnel are imperative.
Surgical Helmet Systems (SHS) are now widely utilized, as they are believed to minimize intraoperative contamination and provide barrier protection to the surgeon. However, the validity of the SHS as a tool to decrease contamination has been called into question, with some studies showing a potential increase in surgical site contamination [11-18]. Unlike the original and more cumbersome Charnley’s negative-pressure Body Exhaust System (BES) [6,8], the SHS consist of an intake fan that blows air around the surgeon’s head and into the remainder of the gown, creating a positive pressure environment [5]. Positive pressure inside the suit may permit air or bacteria to be forced out of any pervious areas, such as the gown cuff. In a recent systematic review, Young et al. [5] reported a ten-fold increase in deep infection rates when SHS were utilized in total hip and knee arthroplasty compared with BES. Moreover, he demonstrated with the use of fluorescent particles, that the Gown Glove Interface (GGI) serves as a potential source of contamination when using SHS.
With the widespread use of SHS in an increasing total joint arthroplasty era, investigational studies reporting on modifiable factors of the susceptible GGI are needed. The purpose of this study is to identify and evaluate the modifiable risk factors that may lead to contamination at the GGI. To our knowledge, no study has analyzed the variables of helmet fan speed, toga waist tightness, and gloving technique in an effort to decrease contamination.
Material and Methods
A pilot study was conducted using a standardized TKA simulation to determine the effects of modifiable factors on contamination using a SHS. All simulations were carried out in a standard operating room utilizing Total Shield Surgical Togas and helmets (Zimmer, Warsaw, IN) and performed on SAWBONES (Vashon Island, Washington). Similar to previous studies [7,11,19,20] fluorescent powder measuring 5-micrometers (Glow Inc., Severn, MD), the typical size of bacterial laden skin particles, was used as a surrogate for shed cells. A total of 12 TKA simulations, each lasting twenty minutes, were conducted utilizing two surgeons.
Prior to gowning and gloving, two surgeons uniformly covered their hands with the fluorescent powder to the level of the wrist crease. In a clean subroom, they were gowned and double-gloved by a trained scrub personnel using standard technique. Care was taken not to contaminate the proximal portion of the glove or gown during the process. The permeable gown cuff was positioned at the level of the metacarpal phalangeal joint in all cases to provide standardization. Surgeons were instructed not to pull the gown sleeve proximally, as is occasionally done for comfort, because this decreases the distance between the permeable gown cuff and proximal GGI. Before starting the simulation an Ultraviolet (UV) light was utilized to ensure no powder contamination occurred during the gowning and gloving process. If powder was noted anywhere on the gown or outside of the glove, both were removed, and the surgeon was redressed until no particles were seen.
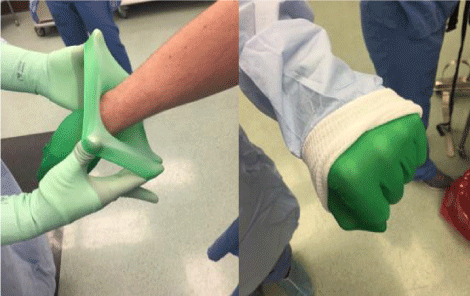
Six different experiments were performed with two different surgeons working simultaneously, resulting in 12 sets of data. Variations of fan speed, waist tightness, and gloving technique were tested as outlined in table 1. The fan speed was determined by the audible tone on the helmet, with “medium” being the middle of the full audible range. A “loose waist” was defined by approximating the very ends of the waist wrap together in the front of the gown. For the “tight waist” variable, the wraps were tightened snug around the surgeon’s torso and tied securely in the front of the gown. The alternate gloving arm of the experiment consisted of the surgeon donning the under-gloves prior to the toga and outer-gloves (Figure 1). This allowed the proximal aspect of the impervious under glove to be well proximal to the permeable gown cuff.
| Table 1: Modifiable parameters for each experiment. | |
| Experiment | Parameters |
| Control | Medium fan speed, loose waist, standard gloving |
| 1 – Low fan | Low fan speed, loose waist, standard gloving |
| 2 – High fan | High fan speed, loose waist, standard gloving |
| 3 – Tight waist | Medium fan speed, tight waist, standard gloving |
| 4- “Worst case” | High fan speed, tight waist, standard gloving |
| 5 – Alternate gloving | Medium fan speed, loose waist, alternate gloving |
Each simulation was conducted for exactly twenty minutes, in which all cuts to the femur and tibia were performed using standard technique and instrumentation. Contamination on the gown was assessed twice during the simulation, once at ten-minutes and again at the completion of the simulation. To assess contamination the lights were turned off and four individuals independently assessed the GGI using a UV light. Contamination was graded using a previously published grading scale 11: 0 (no contamination), 1 (1 to 5 specks), 2 (5 to 10 specks), 3 (10 to 100 specks), and 4 (>100 specks). After assessment of the GGI at the end of twenty minutes, the undersurface of the glove at the GGI was also inspected for contamination. This was graded qualitatively (yes or no) if the proximal undersurface was contaminated. The distance from the wrist crease to the GGI was divided in half and reviewers were asked to identify if particles were present on the proximal portion of this space. The undersurface of the glove at the GGI was defined as the undersurface of the glove that was superficial to the impervious portion of the gown; this was the under-glove and outer-glove in standard and alternate gloving techniques, respectively.
Results
No particle contamination was identified on the outside of the glove for any simulation irrespective of fan speed, toga waist tightness, or gloving technique. However, the undersurface of the glove was contaminated nearing the sterile boundary in all simulations with standard gloving and in no case of alternate gloving.
The control group consisted of medium fan speed, loose waist, and standard gloving to represent the most common subset in clinical practice. Halfway through the control simulation, and again at the completion of the twenty-minutes, each reviewer independently graded the contamination as a 0, indicating no contamination on the gown proximal to the GGI. However, upon review of the glove undersurface all reviewers identified contamination in the proximal half, nearing the sterile boundary.
Helmet fan speed was tested in experimental Groups 1 and 2, low fan speed/loose waist/standard gloving and high fan speed/loose waist/standard gloving, respectively. All reviewers independently graded the contamination on the gown as a 0, representing no contamination proximal to the GGI. However, the undersurface of the glove at the GGI was identified as proximally contaminated in all cases.
Waist tightness was assessed in experimental Group 3, medium fan speed/tight waist/standard gloving. Again, the results were identical to the control group. All reviewers graded GGI contamination as a 0 and identified undersurface contamination proximally.
A theoretical positive pressure “worst case” scenario was also tested, in which high fan speed and a tight waist were tested for particle egress. Even with maximizing the positive pressure within the suit, no reviewer identified any contamination outside the glove at any time point. Glove undersurface contamination was also unchanged from the control and particle egress was present near the sterile boundary proximally.
Consistent with the control group of medium fan speed and loose toga waist, the alternate gloving technique, Group 5, failed to demonstrate contamination outside the glove at all-time points. However, at the completion of the simulation, all reviewers graded no contamination on any portion of the glove undersurface.
Discussion
With previous reports stating that the majority of contamination of total joint arthroplasties occurs intraoperatively, studies that demonstrate a reduction of possible contamination are merited. The GGI has been implicated many times as a potential site for contamination. Ward et al. showed that 27% of direct contact cultures from the surgeon’s volar sleeve were positive one hour into a case [21]. Additionally, gown material has also been implicated in similar studies, with more stiff materials leading to increasing contamination at the GGI [7]. Here, we demonstrate that the commonly utilized alternative gloving technique minimizes contaminate egress at the susceptible GGI compared with standard gloving. With donning of the under-glove prior to the toga, the distance which contaminates from the hand have to travel to breach the sterile boundary is increased. Compared with standard gloving technique, alternate gloving may minimize contamination with the use of positive-pressure SHS.
To maintain consistency within the literature, we performed our study in similar fashion to previous reports [7,11]. For standardization, we used a consistent powder size, timing of mock trial, and means of quantifying contamination. Though previous studies have sought to investigate sources of contamination and differences between gowns, no study has investigated the effects of fan speed, toga waist tightness, or gloving technique as it relates to contamination.
While at the outset of the study, we hypothesized that fan speed and toga waist tightness would have a significant role in contamination, we were surprised to find that these variables seemed to play a minimal role irrespective of gloving technique. Unexpectedly, the gloving technique utilized directly correlated with the degree of contamination on the undersurface of the glove at the GGI. The contamination of the undersurface of the glove at the level of the wrist cuff with standard gloving technique was something we found significant. While we did not witness a frank breech of the GGI as previously reported [7], we did note a rapid migration of fluorescent particles from the hand through the pervious wrist cuff. This area became proximally contaminated with fluorescent particles during each simulation utilizing standard gloving. It stands to reason that with continued motion of the wrists, along with the time required in performing an entire procedure, contamination from the surgeon’s hand to the gown may have been evident. Salvi et al. [20] demonstrated that even after a standard six-minute preoperative hand scrub using povidone-iodine, staphylococcus contaminates were still isolated from the surgeon’s fingers in 50% of cases. Our findings demonstrate that bacterial size particles have a clear communication between the nonsterile hand and the undersurface of the glove at the GGI, representing a potential source for contamination Additionally, it is believed that if the surgeons were allowed to pull the gown sleeve more proximally, as is done for comfort, this would pull gown out from the contaminated glove undersurface and lead to further contamination.
These findings are important as they identify a specific area of contamination with a simple solution. A previous study suggested taping of the GGI with sterile tape as a way to help prevent outflow of contaminates from this weak interface [11]. The gown material would also influence the effectiveness of creating an airtight seal with the taping technique. Here, we demonstrate that an alternative gloving technique results in no powder migration through the permeable cuff and no contamination on the underside of the glove after 20 minutes. Intuitively, with this alternative gloving technique, in order for the powder to reach the sterile field, it would first have to migrate proximally around the cuff of the impervious under-glove, distally to the permeable cuff of the gown, and then proximally again to the cuff of the outer-glove. This provides an added layer of protection to hand contaminates.
While changing the order in which gloves are donned seems to be a simple solution, it is not without difficulties. First, this technique is predicated on the ability of the scrubbed personnel to follow proper technique. For alternate gloving, once the surgeon’s hand is in the glove, the glove cuff must be quickly released to prevent contamination of the scrub tech from the surgeon’s un-gowned forearms. Additionally, if gloves are to be changed intraoperatively, as is occasionally done prior to handling implants, the relationship of the permeable cuff to the impermeable under glove must be maintained. Alternatively, the surgeon has the option of donning a third pair of gloves prior to handling implants.
There are several limitations to our study. First, the type of toga system utilized has not been previously studied. As this toga is the one used at our facility, its vulnerability to contamination of the GGI is speculative as there is no study for which to compare our data. However, the authors feel that the material is comparable to most toga systems and this likely had no influence on the findings between alternative gloving techniques as it was kept constant through all experiments. Also, this technique only addresses contamination of the hand and distal forearm and does not consider contamination from other parts of the body. Since the cuff is the only permeable part of the gown, we feel this to be the most vulnerable area for contamination. Finally, our study consisted of only twelve data points, two of which utilized the alternate gloving technique. We believe that this alternate gloving technique, if done properly, could considerably increase the sterile boundary, and therefore, decrease contamination potential from the hand at the GGI. Additional studies are needed to further evaluate the relationship between gloving technique and contamination that has been identified in the present study.”
Conclusion
Modern surgical toga systems have been shown to increase infection rates when compared to standard surgical gowns. The use of a commonly practiced alternative gloving technique, utilizing the placement of the under-glove prior to the gown may result in less contamination of the undersurface of the outer-glove and at the GGI.
References
- Kurtz SM, Lau E, Watson H, Schmier JK, Parvizi J. Economic burden of periprosthetic joint infection in the United States. J Arthroplasty. 2012; 27: 61-65. https://goo.gl/Av2bou
- Namba RS, Paxton L, Fithian DC, Stone ML. Obesity and perioperative morbidity in total hip and total knee arthroplasty patients. J Arthroplasty. 2005; 20: 46-50. https://goo.gl/nJSJKY
- Kurtz S, Ong K, Lau E, Mowat F, Halpern M. Projections of primary and revision hip and knee arthroplasty in the United States from 2005 to 2030. J Bone Joint Surg Am. 2007; 89: 780-785. https://goo.gl/2tUviy
- Bozic KJ, Kurtz SM, Lau E, Ong K, Chiu V, Vail TP, et al. The epidemiology of revision total knee arthroplasty in the United States. Clin Orthop Relat Res. 2010; 468: 45-51. https://goo.gl/UdWXfE
- Young SW, Zhu M, Shirley OC, Wu Q, Spangehl MJ. Do 'Surgical helmet systems' or 'Body exhaust suits' affect contamination and deep infection rates in arthroplasty? A systematic review. J Arthroplasty. 2016; 31: 225-233. https://goo.gl/v8CA1i
- Charnley J. A Clean-Air operating enclosure. Br J Surg. 1964; 51: 202-205. https://goo.gl/FGWHnS
- Fraser JF, Young SW, Valentine KA, Probst NE, Spangehl MJ. The Gown-glove interface is a source of contamination: a comparative study. Clin Orthop Relat Res. 2015; 473: 2291-2297. https://goo.gl/enkVMf
- Lidwell OM, Lowbury EJ, Whyte W, Blowers R, Stanley SJ, Lowe D. Effect of ultraclean air in operating rooms on deep sepsis in the joint after total hip or knee replacement: a randomised study. Br Med J (Clin Res Ed). 1982; 285: 10-14. https://goo.gl/vkEVgE
- Owers KL, James E, Bannister GC. Source of bacterial shedding in laminar flow theatres. J Hosp Infect. 2004; 58: 230-232. https://goo.gl/qADfy5
- Whyte W, Hodgson R, Tinkler J. The importance of airborne bacterial contamination of wounds. J Hosp Infect. 1982; 3: 123-135. https://goo.gl/DQFzMD
- Young SW, Chisholm C, Zhu M. Intraoperative contamination and space suits: a potential mechanism. Eur J Orthop Surg Traumatol. 2014; 24: 409-413. https://goo.gl/io5vQ8
- Hooper GJ, Rothwell AG, Frampton C, Wyatt MC. Does the use of laminar flow and space suits reduce early deep infection after total hip and knee replacement? The ten-year results of the New Zealand Joint Registry. J Bone Joint Surg Br. 2011; 93: 85-90. https://goo.gl/5TdHDE
- Bohn WW, McKinsey DS, Dykstra M, Koppe S. The effect of a portable HEPA-filtered body exhaust system on airborne microbial contamination in a conventional operating room. Infect Control Hosp Epidemiol. 1996; 17: 419-422. https://goo.gl/wTS4hE
- Pasquarella C, Pitzurra O, Herren T, Poletti L, Savino A. Lack of influence of body exhaust gowns on aerobic bacterial surface counts in a mixed-ventilation operating theatre. A study of 62 hip arthroplasties. J Hosp Infect. 2003; 54: 2-9. https://goo.gl/GA7xFv
- Shaw JA, Bordner MA, Hamory BH. Efficacy of the Steri-Shield filtered exhaust helmet in limiting bacterial counts in the operating room during total joint arthroplasty. J Arthroplasty. 1996; 11: 469-473. https://goo.gl/dBcP2D
- Der Tavitian J, Ong SM, Taub NA, Taylor GJ. Body-exhaust suit versus occlusive clothing. A randomised, prospective trial using air and wound bacterial counts. J Bone Joint Surg Br. 2003; 85: 490-494. https://goo.gl/p9TM6Y
- Namba RS, Inacio MC, Paxton EW. Risk factors associated with surgical site infection in 30,491 primary total hip replacements. J Bone Joint Surg Br. 2012; 94: 1330-1338. https://goo.gl/Rbnav8
- Namba RS, Inacio MC, Paxton EW. Risk factors associated with deep surgical site infections after primary total knee arthroplasty: an analysis of 56,216 knees. J Bone Joint Surg Am. 2013; 95: 775-782. https://goo.gl/bGEcB5
- Clark RP, Reed PJ, Seal DV, Stephenson ML. Ventilation conditions and air-borne bacteria and particles in operating theatres: proposed safe economies. J Hyg (Lond). 1985; 95: 325-335. https://goo.gl/DxsoKY
- Salvi M, Chelo C, Caputo F, Conte M, Fontana C, Peddis G, et al. Are surgical scrubbing and pre-operative disinfection of the skin in orthopaedic surger reliable? Knee Surg Sports Traumatol Arthrosc. 2005; 14: 27-31. https://goo.gl/gmYQkZ
- Ward WG, Cooper JM, Lippert D, Kablawi RO, Neiberg RH, Sherertz RJ. Glove and gown effects on intraoperative bacterial contamination. Ann Surg. 2014; 259: 591-597. https://goo.gl/ddSdZ2
Authors submit all Proposals and manuscripts via Electronic Form!