The Phenomenon of Weak, Unawakening afferent Signals Involvement in Synchronization Mechanisms of Neuron activity during delta Sleep
Shakhnarovich V M, Indursky P A, Dementienko V V*, Markelov V V
JSC “NEUROCOM”, Moscow, Russia
*Address for Correspondence: Dementienko V V, Kotelnikov Institute of Radio Engineering and Electronics, Russian Academy of Sciences, Moscow, Russia, E-mail: valery@dementienko.ru/ valery_v_d@mail.ru
Submitted: 22 August 2017; Approved: 02 October 2017; Published: 04 October 2017
Citation this article: Shakhnarovich VM, Indursky PA, Dementienko VV, Markelov VV. The Phenomenon of Weak, Unawakening afferent Signals Involvement in Synchronization Mechanisms of Neuron activity during delta Sleep. Int J Sleep Disord. 2017;1(1): 007-011.
Copyright: © 2017 Shakhnarovich VM, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited
Download Fulltext PDF
Changes in sleep characteristics were studied under the non-wake-up stimulation with current pulses of less than 1 μA on average, applied to the palmar surface skin receptors during delta-sleep. A significant increase in duration of the first and second cycles of deep sleep has been found, as well as a shorter latent period before the delta-sleep onset and a longer time of the Rapid Eyes Movement Sleep (REM). The sleep structure improvement was accompanied by the reduced reactive anxiety and depression and an increase in subjective physical efficiency.
Introduction
According to WHO, sleep disorders affect up to 15% of the world population. In developed countries, this figure is substantially higher. In most cases, sleep disturbance is connected with the functional disorders of the central nervous system and with depression, which is now rather well-studied [1-3].
Sleep studying in patients with functional disorders of the central nervous system [2] showed the following changes in a sleep pattern: a decrease in deltaindex; reduced duration of the entire slow-wave sleep stage, especially in the first and second sleep cycles; elongated latent period of the second to third stage transition (from the first sleep spindle to the first deltawave) ; the reduced latent period of the Rapid Eye Movement (REM) phase onset and a greater REM representation in the first half of the night. With improvement of the patient functional state, a reverse dynamics of these parameters occurs [4, 5].
We suggested that not only a direct dependence observed in treatment of depression exists [the condition improvement is accompanied by improvement of sleep [6]], but another statement is true, namely, improvement of the sleep structure is followed by improvement of the patient condition [7]. Thus, an effective method of the sleep structure correction may be expected to be useful for elimination of the reactive anxiety and depression. In this study, we aimed at finding of a physical nondrug way of changing the sleep structure and developing of a method for the sleep quality improvement.
Materials and Methods
First, we have selected a sub threshold electrical stimulation from several non-wake-up actions of various modalities. The effect was the most pronounced when the patients were stimulated at the slow-wave sleep stage (delta-sleep).
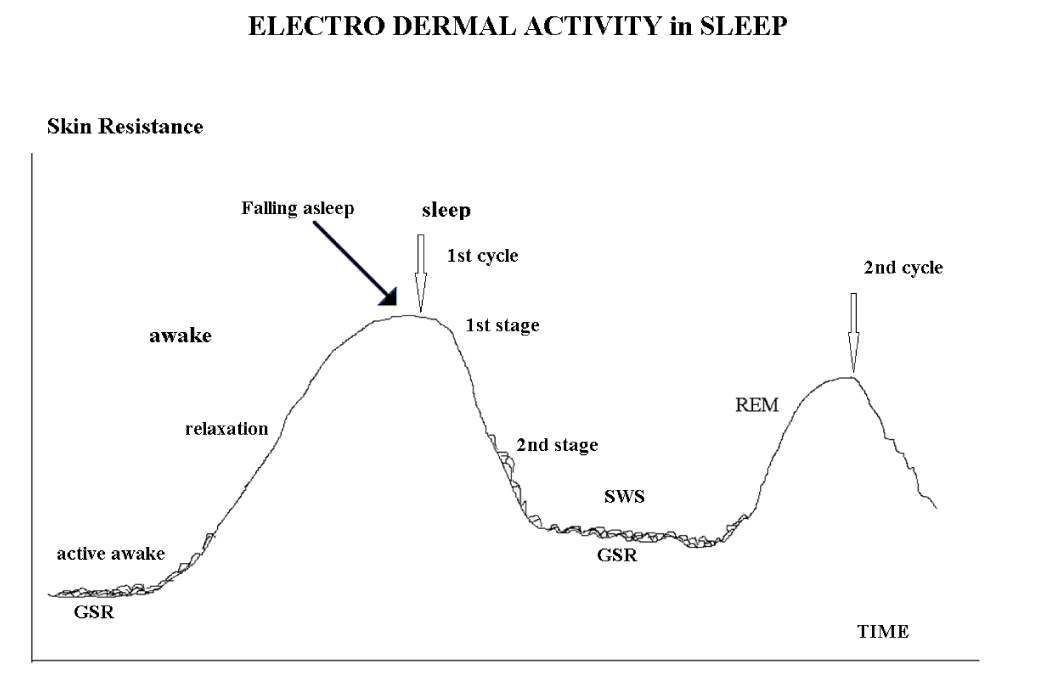
For SWS (Slow-Wave Stages) identification we have used well-known Electro Dermal Activity [EDA] feature [8, 9] which is defined by its tonicity values and phasic parameters Galvanic Skin Response [GSR] (Figure 1]. Specifically this EDA feature we used for sleep stages control during delta sleep stimulation in domestic conditions [10].
The therapeutic treatment was a sub threshold electrical stimulation of the palmar surface skin receptors by the current pulses up to 100 μA repeated with frequency of 0.5-1.2 Hz for 3-10 ms.
Our study involved 57 subjects (32 men and 25 women) aged from 27 to 55 years who were discontent with sleep and complained about bad mood and working capacity. A control group involved 19 subjects 26-37 years of age who also were complaining about sleep disorders.
Examination of each subject passed through five steps
Step 1
• Survey was conducted using standard sleep questionnaires.
• Psychological testing. In order to identify depression symptoms manifestation degree and reactive anxiety level, tests of Tailor Manifest Anxiety Scale [12], Spielberger State Anxiety Inventory [13] and Beck Depression Inventory [14] were used.
• Blood pressure was measured.
• Laboratory polysomnographic studies of the night sleep without stimulation for two nights using standard poly somnography with additional electro-dermal activity dynamic registration [10].
Step 2
• Poly graphic studies of the night sleep in the laboratory, similarly to Step 1, with stimulation during the deltastage of sleep for one night.
Step 3
• Home sleep with a device that stimulates a subject during delta sleep for seven consecutive nights. Diagnosing of sleep stages and stimulation during the slow-wave sleep phases were conducted according to the biofeedback scheme by measurement of the electro dermal activity [10].
Preliminary studies of efficiency showed that the primary fixation of the therapeutic effect occurred on the six to seven days of the nocturnal stimulation.
Step 4
• Sleep with a device in the laboratory under the polygraphic control for one night.
Step 5
• Final questioning,
• Psychological testing
• The blood pressure measurement.
To assess the subjects’ state, polysomnographic analysis and psychological testing were used: Beck test, Taylor scale, Spielberger test, and WAM technique [self-reports about well-being, activity, and mood]. The results were processed statistically using Student’s t test. The differences were considered significant at p < 0.05.
Results
Analysis of our results showed that in all subjects, the delta sleep duration increased significantly, after the seven-day stimulation: on average, by a factor of 1.3 (p = 0.04).
This effect was observed in 60% of all 57 test subjects. Note that in a group of subjects who had a longer deltasleep already in the first night with stimulation, this parameter increased by a factor of 1.6 on average (p = 0,0005) after the complete course and the effect was observed in 85% of subjects of this group. Thus, we used an objective criterion of patient selection; therefore, this is a promising method of the sleep quality improvement.
Delta sleep duration, min
The use of this criterion enabled us to identify in the background experiments [the first night] a group of subjects with significantly shorter delta-sleep duration than in the entire sample on average [34 subjects out of 57 test subjects, [Figure 2]. In other words, we were dealings with the objective sleep problems of a certain group of subjects.
Improvement of the sleep quality after a stimulation course (Figure 2) was also confirmed by a 1.3-fold shortening of the delta-sleep latent period (the time between the first sleep spindle and the first delta wave of at least 70 μV), as well as by a 1.4-fold increase in the rapid sleep latent period and a significant 1.5-fold decrease in the average duration of the first Rapid Sleep Episode [REM]. In addition, in the slow-wave phase, the power of delta activity and the delta wave average amplitude increased significantly. In other words, according to the old classification, the fourth stage was significantly more pronounced [at least 50% of delta waves per epoch].
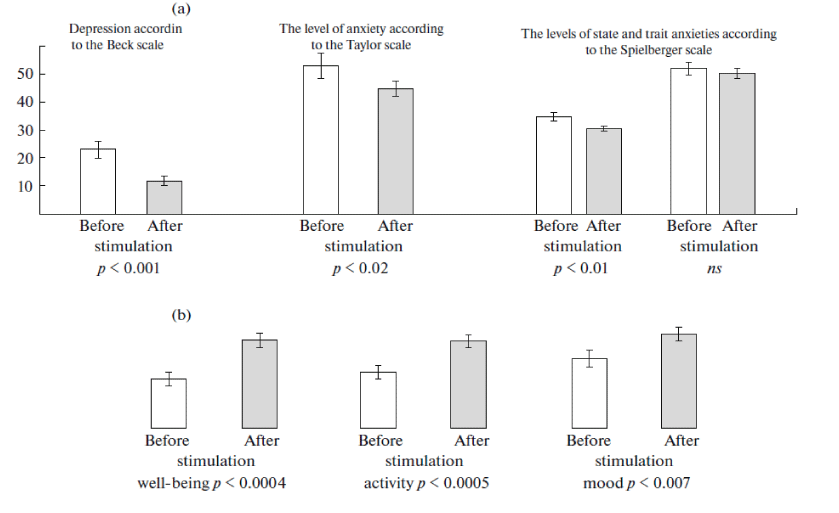
The final stage of psychological testing [Figure 3] showed that, after electro stimulation, depression of the test subjects and the level of their reactive anxiety were significantly reduced as compared to the background parameters. In the morning after sleep, these subjects were in a good mood and had a subjective state of having a good night sleep and better working efficiency.
Analysis of the psychological state dynamics showed that the condition improvement was similar in the group selected according to the above criterion and in all test subjects as a whole. Probably, the use of a device for stimulation causes a suggestive effect, which in itself is positive and does not contradict a lengthening of the test subjects’ deltasleep.
After a stimulation course, in the test subjects, the level of systolic Blood Pressure (BP) was reduced on average by 20 mm Hg. This BP dynamics was accompanied by a decrease in the level of anxiety and reactive depression.
There were no any significant changes in sleep characteristics as compared to the background parameters in the control subjects. However, some of them [six subjects] had several positive changes in sleep characteristics as well as they were feeling better [WAM technique], which was clearly indicative of the placebo component in our technology.
The results obtained may be probably explained by a phenomenon of sleep protection against the weak centripetal wake-up pulses from skin receptors. This phenomenon is induced by a neurophysiologic mechanism that activates sub cortical structures responsible for the cortical neuron synchronization in the delta activity regime. Passing of the wake-up pulses via reticular formation and, probably, via the thalamus is blocked completely or partially. The process is referred to as the mechanism of involvement.
Probably, this mechanism underlies correction of the slow-wave sleep by means of electrical stimulation, which is accompanied by improvement of general psychological condition of the test subjects due to processes occurring in the delta sleep. During this very stage, the anterior lobe of pituitary gland is known to produce at least two-thirds of the somatotropic hormone responsible for protein synthesis, while epiphysis is known to produce melatonin, the hormone of good mood, performance, and memory. At the same time, memorization processes are consolidated, which is interpreted by patients as improvement of self-rated well-being and working efficiency.
In our previous publication [7] we voiced the proposal that unawaking incentives physical modality can vary. Therefore, involvement mechanism appears to be universal. This data supports the outcome of the research [15] where acoustic stimulation was used. Either one of the outcomes [7,15] open significant opportunities for creation of automated means of SWS correction and insomnia therapy.
Acknowledgments
The study was supported by the Russian Foundation for Basic Research, project no. 14-07-00580.
- Rotenberg, V.S, The Adaptive Function of Sleep and the Causes and Manifestations of Its Disturbance, [in Russian]. Moscow: Nauka, 1982.
- Buysse, D.J., Insomnia. Principles and Practice of Sleep Medicine, Eds:Kryger M.H., Roth, T., and Dement, Elsevier, New York, 2005, pp. 702–760.
- Koval’zon, V.M., Principles of Somnology [in Russian], Moscow: BINOM, 2011.
- Massimini M, Ferrareli F, Esser, S.K, Riedner BA, Huber R, Murphy M, et al. Triggering Sleep Slow Waves by Transcranial Magnetic Stimulation. Proc. Natl. Acad. Sci. U.S.A. 2007; 104 : 8496–8512. https://goo.gl/H17fJo
- Indursky P. A New Application of rTMS: Sleeping Brain and Depression. Med Hypotheses. 2001; 57: 91–92. https://goo.gl/StTzM2
- Cohrs, S., Tergau, F., Riech, S., Kastner, S., Paulus, W., Ziemann, U., Ruther, E., and Hajak, G., High-Frequency Repetitive Transcranial Magnetic Stimulation Delays Rapid Eye Movement Sleep. Neuroreport. 1998; 9 : 3439–3443. https://goo.gl/X4ZrgM
- Indursky P.A, Markelov V.V, Shakhnarovich V.M, and Dorokhov V.B. Low-Frequency Rhythmic Electrocutaneous Hand Stimulation during Slow Wave Night Sleep. Human Physiol. 2013; 39 : 642–654. https://goo.gl/JfqW3q
- Savchenko, V.V., Biological Feedback and Technological Training of Operators with Use of Tremor Parameters, (in Russian). Sist. Analiz Upravl. Biomed. Sist,2006, vol. 5, no. 1, pp. 187–191.
- Rotenberg V.S., Kochubey B.I., Shakhnarovich V.M. The Dynamics of Electro Dermal Activity in Different Night Sleep Stages and Cycles. Zh Vyssh Nerv Deiat I P Pavlova
- Indursky P.A, Dunaevsky L.V, Shakhnarovich V.M, Dementienko V.V. A method for correcting the state of patients with sleep disorders by electrostimulation based on recording electrodermal activity. RF Patent 2 431508, 2010.
- Bastien C.H, Vallieres A, Morin C.M, Validation of the insomnia severity index as an outcome measure for insomnia research. Sleep Medicine. 2001; 2: 297-307. https://goo.gl/gMChKX
- Taylor J.A. A Personality Scale of Manifest Anxiety. J. AbnormPsychol. 1953; 48 : 285-290. https://goo.gl/P7mkhG
- Spielberger, C. D. (1989). State-Trait Anxiety Inventory: Bibliography (2nd ed.). Palo Alto, CA: Consulting Psychologists Press.
- Beck AT. Depression: Causes and Treatment. Philadelphia: University of Pennsylvania Press.1972; 281-282.
- Ngo HV, Miedema A, Faude I, Martinetz T, Molle M, Born J. Driving Sleep Slow Oscillation by Auditory Closed-Loop Stimulation. Neurosci.2015 ; 35 : 6630–6638. https://goo.gl/1bXW9q

Sign up for Article Alerts