H-Point Assay Method for Simultaneous Determination of Paracetamol and Diclofenac Sodium in Their Combined Pharmaceutical Dosage Forms
Mahmoud M. Sebaiy1* and Amr A. Mattar2
1Medicinal Chemistry Department, Faculty of Pharmacy, Zagazig University, Zagazig 44519, Egypt
2Pharmaceutical Medicinal Chemistry Department, Faculty of Pharmacy, Egyptian Russian University, Badr City, Cairo 11829, Egypt
*Address for Correspondence: Mahmoud M. Sebaiy, Medicinal Chemistry Department, Faculty of Pharmacy, Zagazig University, Zagazig 44519, Egypt, E-mail: mmsebaiy@zu.edu.eg/ sebaiym@gmail.com
Submitted: 09 December 2019; Approved: 17 January 2020; Published: 21 January 2020
Citation this article: Sebaiy MM, Mattar AA. H-Point Assay Method for Simultaneous Determination of Paracetamol and Diclofenac Sodium in Their Combined Pharmaceutical Dosage Forms. Open J Biotechnol Bioeng Res. 2020;4(1): 001-005.
Copyright: © 2020 Sebaiy MM, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited
Keywords: Spectrophotometric; Paracetamol; Diclofenac sodium; H-Point assay; ICH guidelines
Download Fulltext PDF
A simple, specific, accurate and precise spectrophotometric method were settled for simultaneous determination of paracetamol and diclofenac sodium in pure form and in their pharmaceutical formulation. H-Point assay has been used in simultaneous determination of both drugs without prior separation. H-Point assay method parameters were validated according to ICH guidelines in which accuracy, precision, repeatability and robustness were found in accepted limits. Advantages and disadvantages of H-point assay were discussed and statistical comparison between the proposed method and the reference method was performed.
Introduction
Paracetamol (PAR); N-(4-Hydroxyphenyl)acetamide (Figure 1) is related to NSAID (Non-Steroidal Anti-Inflammatory Drugs) which can act both centrally and peripherally for the treatment of non-inflammatory conditions in patients having gastric symptoms [1].
Diclofenac sodium (DCL); 2-(2,6-dichloroamino)phenylacetic acid (Figure 1) [2], is an analgesic and anti-inflammatory agent which can act by the inhibition of the synthesis and the release of leukotrienes and prostaglandins [3]. DCL can be combined with PAR as the latter can provide basic relief before DCL and enhances its pain-relieving and antipyretic effect. PAR & DCL combination can be used in treatment of several diseases not only inflammation [4].
The literature demonstrated that several methods were accomplished for the analysis of PAR and DCL in their mixture form or in combination with other drugs. PAR & DCL have been determined by spectrophotometric methods [5-9], HPLC methods [10-13], TLC methods [14,15], capillary zone electrophoresis method [16], voltammetric method [17], a method based on poly (diallyldimethylammonium chloride) functionalized graphene [18] and a method based on Au nanoparticles - functionalized graphene/poly (L-Arginine) glassy carbon electrode [19]. To the best of our knowledge, there is no reported method for the determination of this drug mixture using H-point assay technique. As such, the aim of work is to develop a spectrophotometric method which is accurate, fast and non-complicated for determination of PAR & DCL combination without the interference of their additives or their excipients in pharmaceutical formulations.
Experimental
Apparatus
JASCO dual beam (Japan) UV-visible spectrophotometer model V-630, connected to an ACER compatible computer with spectra manager II software was used. The spectral slit width is 2 nm at speed up can be increased up to 8000 nm/min. All the measurements have been carried out in 1 cm quartz cell. The wavelength ranges were 200-400 nm at room temperature. Also, PASW statistics 18® software program was used for statistical analysis.
Materials and reagents
Pure standards: PAR and DCL were kindly provided by EIPICO (Egypt). Their purity was claimed to be as 99.50% and 99.80 % for PAR & DCL, respectively.
Pharmaceutical formulations: Diclocin® tablets were purchased from the market (label claim: PAR 250 mg + DCL 50 mg) produced by Cipcopharmaceuticals, India.
Solvents: HPLC grade Methanol was purchased from LiChrosolv, Merck KGaA (Germany). All of measurements have been accomplished by using 90% Methanol.
Standard solutions: Standard stock solutions (1 mg/mL) of PAR and DCL were prepared in 90% methanol. Working standard solution of PAR (40 µg/mL) and DCL (50 µg/mL) were prepared by further dilution with 90% methanol.
Laboratory prepared mixtures: Different ratios of PAR & DCL were performed by transferring aliquots from their standard solutions to volumetric flasks (10 mL) and then dilution was carried out with 90% methanol.
Procedures
Construction of calibration curves
For PAR: Working solutions equivalent to 4-22 µg/mL were prepared by addition of aliquots (1, 1.50, 2, 2.50, 3, 3.50, 4, 4.50, 5, 5.50 mL) of PAR working standard solution (40 µg/mL) to 10 mL volumetric flasks followed by dilution with 90% methanol.
For DCL: Working solutions equivalent to 5-45 µg/mL were prepared by adding aliquots (1, 1.50, 2, 2.50, 3, 3.50, 4, 4.50, 5, 6, 7 mL) of DCL working standard solution (50 µg/mL) to 10 mL volumetric flasks followed by dilution with 90% methanol. Measurements of the absorption spectra were carried out at room temperature over the wavelengths (200-400 nm).
H-Point assay method
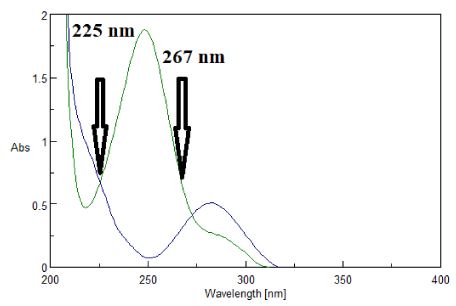
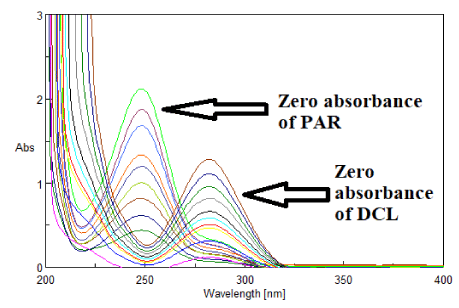
In the proposed method, two wavelengths were selected, 225 nm and 267 nm at which PAR exhibited the same absorptivity, in contrast to DCL that demonstrated sufficient difference in absorptivity at each of the selected wavelengths as shown in figure 2. Two calibration curves were constructed for PAR at 225 nm and 267 nm and its zero absorbance spectrum is showed in figure 3.
On the other hand, DCL was the component, from which standard solutions of increased concentration could be added to a mixture of both components to determine the concentration of both by H-point standard addition method.
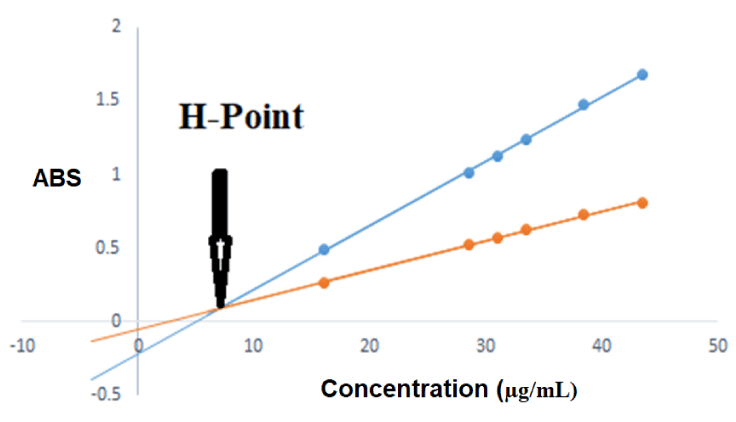
Aliquots containing 7.5, 12.5, 17.5, 20, 22.5, 25, 30, 35 µg/mL DCL were accurately added to the laboratory prepared mixture or pharmaceutical dosage form prepared solutions to be determined by this method. Curves of standard addition method were constructed where absorbances of solutions after DCL standard addition were represented on Y-axis while concentrations of the added DCL standard were represented on X-axis at the two selected wavelengths (225 nm and 267 nm). By plotting the absorbance versus added DCL concentration, two straight lines of different slopes and intercepts were obtained. As the absorbance value of PAR is constant at the two selected wavelengths (225 nm and 267 nm), all straight lines obtained at the different wavelengths by applying the standard additions method will have a common point. This point is known as the H point (Figure 4), the abscissa refers to DCL concentration (C DCL) alone and the Y coordinate is the absorbance of PAR (A PAR) alone in the corresponding mixture. The concentration of PAR (C PAR) in mixture was then determined by substitution in any of regression line equations at 225 nm and 267 nm.
Analysis of laboratory prepared mixtures
The spectra of the mixtures were measured after preparation of different ratios of the laboratory prepared mixtures then handled in the same conditions as described under each method.
Application to pharmaceutical formulation
10 tablets of Diclocin® were weighed and crushed then an amount equivalent to 50 mg PAR and 10 mg DCL in each tablet was transferred into a volumetric flask (50 mL) and diluted with 90% methanol as follow: First, 30 mL of 90% methanol were added and sonicated then dilution was carried out to the mark and filtered. Second, 10 mL of the dilution was transferred into a 100 mL volumetric flask to give a concentration equivalent to 100 µg/mL PAR and 20 µg/mL DCL. Third, any further dilutions were carried out in volumetric flasks (10 mL) and treated in the same way as described under each method.
Results and Discussion
Method optimization
Two major problems were found during the analysis of PAR & DCL binary mixture; first, the overlapped spectra between the absorptivity of both drugs, and second, PAR, the main (major) constituent, had unfortunately very high absorbance, while DCL, the minor component, had low absorbance value. Intrinsically, sample enrichment technique [20] was used in which the concentration of DCL (the minor component) in their dual mixtures was increased to facilitate its determination. This was carried out by adding a fixed amount of standard DCL to each experiment when combined with PAR, then subtraction of its concentration before the calculation of the required concentration of DCL. Sample enrichment technique has been used for solving the same problem in the analysis of other drug mixtures of different drug ratios [21,22].
H-Point assay method
The determination of concentration of drug X using H-point assay method requires the selection of two wavelengths λ1 and λ2, at which the interferent species, Y, has the same absorbance. Then, known amounts of X are successively added to the mixture and the resulting absorbances are measured at the two wavelengths and expressed by the following equations:
A(λ1) = b0 + b +Mλ1Ci (1)
A(λ2) = A0 + A +Mλ2Ci (2)
Where, A(λ1) and A(λ2) are the analytical signals measured at λ1 and λ2, respectively. b0 and A0 (b0 and A0 are not equal) are the original analytical signal of X at A(λ1) and A(λ2), respectively.
b and A’ are the analytical signals of Y at A(λ1) and A(λ2), respectively. Mλ1 and Mλ2 are the slopes of the standard addition calibration lines at λ1 and λ2, respectively and Ci is the added X concentration. The two straight lines obtained intersect at the so-called H-point (-CH, AH).
At H-point, since A(λ1) =A(λ2), Ci=CH, from Eqs. (1) and (2) it follows that:
b0 + b +Mλ1 (-CH) = A0 + A’ +Mλ2 (-CH) (3)
−CH = [(A0 − b0) + (A’− b)]/ (Mλ1−Mλ2) (4)
225 and 267 nm absorbances were used for determination of PAR & DCL in presence of each other at the same wavelengths. The calibration curves revealed accepted linear relationships between concentrations and absorbance in a range of 4-22 µg/mL for PAR and 7.50-45 µg/mL for DCL with correlation coefficients of ≥ 0.9990 for both drugs. The accuracy of the method illustrated accepted values with 100.25% ± 1.29 for PAR and 100.32% ± 0.51 for DCL. The specificity of the methods demonstrated accepted values with 100.32% ± 0.51 for PAR and 100.25% ± 1.29 for DCL. The results are detailed in table 1.
H-Point assay is very easy and simple as it depends on zero absorption spectra without the need of extra processing. On the other hand, it has two limitations; which are the need for some specific calculations to determine the values of H-Point in addition to requiring more time for performing the standard addition on each mixture.
Method validation
All methods were legalized as demonstrated by ICH guidelines [23]. The linear regression data for the calibration curves showed good linear relationships (Table 1). The accuracy was assessed by analyzing the standard addition method where satisfactory results were achieved as shown in (Tables 1,2). The specificity of this technique was assessed by assaying the laboratory prepared mixtures of PAR & DCL within the linearity range and good results were obtained (Table 1). The intra and inter-day precisions were computed by the analysis of 3 different concentrations of the drugs 3 times on the same day in addition to 3 successive days (Table 1).
Application to pharmaceutical formulation
H-point assay method was successfully applied for determination of PAR & DCL in its pharmaceutical formulation (Diclocin® tablets) the results were acceptable in agreement with the labelled quantities. The standard addition method was used and revealed that no interference of the excipients was observed (Table 2).
Statistical analysis
Statistical comparison between the proposed technique and the reference method was done by One-way ANOVA method through utilizing PASW statistics 18® software program. The calculated F values were less than the theoretical ones indicating that there was no significant difference between them (Table 3).
Conclusion
H-Point assay method was successfully applied for the determination of paracetamol and diclofenac sodium in their binary mixtures and in their dosage form. The proposed method is simple, sensitive and accurate and could be used for routine analysis by using simple technology or instruments. By comparison with the previous reported methods, it was concluded that H-point assay method doesn’t require extra processing but it needs more time and calculations. Statistical comparison revealed that there is no observed significant difference between the proposed method and the reference one.
- Yehia AM, Abd El-Rahman MK. Application of normalized spectra in resolving a challenging Orphenadrine and paracetamol binary mixture. Spectrochim Acta A Mol Biomol Spectrosc. 2015; 138: 21-30. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/25437841
- Abdul Barry M Mahood, Mohammed J Hamezh. Spectrophotometric determination of diclofenac sodium in pharmaceutical preparations. Journal of Kerbala University. 2009; 7: 1-7. http://bit.ly/2u76pSV
- Scholer DW, Boettcher I, Ku EC, Schweizer A. Pharmacology of diclofenac sodium (Voltaren®). Seminars in Arthritis and Rheumatism. 1985; 15: 61-64. http://bit.ly/2NAUwvd
- Brogden RN, Heel RC, Pakes GE, Speight TM, Avery GS. Diclofenac sodium: A review of its pharmacological properties and therapeutic use in rheumatic diseases and pain of varying origin. Drugs. 1980; 20: 24-48. PubMed: https://www.ncbi.nlm.nih.gov/pubmed/6772422
- Saheb DJ, Reddy NR, Chakravarthy IE. Simultaneous determination of paracetamol and diclofenac sodium from combined dosage forms by absorbance difference method. Asian Journal of Chemistry. 2004; 16: 767-772.
- Hegazy MA, Elshahed MS, Toubar SS, Helmy MI. Efficient processing of single and multiple spectral variables for resolution and quantitation of paracetamol, chlorzoxazone and diclofenac. Journal of Advanced Pharmacy Research. 2018; 2: 269-282. http://bit.ly/2Tv3gqr
- Phaneemdra D, Nagamalleswari G. Quantitative analysis of paracetamol and diclofenac in combined dosage form by first derivative and simultaneous equation method in application to the determination of dissolution study. International Journal of Pharmaceutical Sciences and Research. 2012; 3: 3871-3876. http://bit.ly/2u8BLZ4
- Sharma R, Pathodiya G, Mishra GP, Sainy J. Spectrophotometric methods for simultaneous estimation of paracetamol and diclofenac sodium in combined dosage form by application of hydrotropic solubilization. Journal of Pharmaceutical Sciences and Research. 2010; 2: 821-826. http://bit.ly/3amBSkK
- Sebaiy MM, El-adl SM, Mattar AA. Spectrochimica ActA PARt A: Molecular and biomolecular spectroscopy different techniques for overlapped UV spectra resolution of some co-administered drugs with paracetamol in their combined pharmaceutical dosage forms. Spectrochimica ActA PARt A: Molecular and Biomolecular Spectroscopy. 2020; 224: 117429. http://bit.ly/2Rpgy59
- Walash MI, Ibrahim F, Abo S, Abass E. Development and validation of HPLC method for simultaneous estimation of famotidine, paracetamol and diclofenac in their raw materials and pharmaceutical formulation. Analytical Chemistry Letters. 2017; 7: 421-437. http://bit.ly/2Txbm1L
- Jana K, Adhikari L, Moitra S, Behera A. Analysis of multicomponent drug formulations (Diclofenac and paracetamol). Asian Journal of Pharmaceutical and Clinical Research. 2011; 4: 2-4. http://bit.ly/38bO7yz
- Badgujar MA, Pingale SG, Mangaonkar KV. Simultaneous determination of paracetamol, chlorzoxazone and diclofenac sodium in tablet dosage form by high performance liquid chromatography. Journal of Chemistry. 2011; 8: 1206-1211. http://bit.ly/2Nzbiuy
- Nayak VG, Bhate VR, Purandare SM, Dikshit PM, Dhumal SN, Gaitonde CD. Rapid liquid chromatographic determination of paracetamol and diclofenac sodium from a combined pharmaceutical dosage. Drug Development and Industrial Pharmacy. 1992; 18: 369-374. http://bit.ly/364N95E
- Dighe VV, Sane RT, Menon SN, Tambe HN, Pillai S, Gokarn VN. Simultaneous determination of diclofenac sodium and paracetamol in a pharmaceutical preparation and in bulk drug powder by high-performance thin-layer chromatography. Journal of Planar Chromatography. 2006; 19: 443-448. http://bit.ly/3aihhxQ
- Mohammad ALI, Sharma S. Separation of Co-existing paracetamol and diclofenac sodium on silica gel “H” layers using surfactant mediated mobile phases: Identification of diclofenac sodium from human urine. Farmacia. 2009; 57: 201-211. http://bit.ly/2QZxoJ0
- Solangi A, Memon S, Mallah A, Memon N. Determination of ceftriaxone, ceftizoxime, paracetamol, and diclofenac sodium by capillary zone electrophoresis in pharmaceutical formulations and in human blood. Turk J Chem. 2010; 34: 921-933. http://bit.ly/363Nci0
- Guadalupe YAL, Juan MGS, Alberto RH, Jose ARA, Maria EPH, Giaan AAR. Voltammetric determination of diclofenac in the presence of paracetamol and naproxen by an artificial neural network model using a carbon paste electrode. ECS Transactions. 2018; 84: 195-205. http://bit.ly/2NBALUe
- Okoth OK, Yan K, Liu L, Zhang J. Simultaneous electrochemical determination of paracetamol and diclofenac based on poly (diallyldimethylammonium chloride) functionalized graphene. Electroanalysis. 2016; 28: 76-82. http://bit.ly/2tuzqb9
- Esmail Afshar, Fahimeh Jalali. Sensitive simultaneous determination of paracetamol and diclofenac based on au nanoparticles - functionalized graphene/poly (L-arginine) glassy carbon electrode. Journal of the Chilean Chemical Society. 2016; 61: 2846-2851. http://bit.ly/2TwW0uo
- Lotfy HM, Tawakkol SM, Fahmy NM, Shehata MA. Successive spectrophotometric resolution as a novel technique for the analysis of ternary mixtures of pharmaceuticals. Spectrochimica ActA PARt A: Molecular and Biomolecular Spectroscopy. 2014; 121: 313-323. http://bit.ly/2tqw5K8
- Moussa BA, Mahrouse MA, Fawzy MG. Different resolution techniques for management of overlapped spectra: Application for the determination of novel co-formulated hypoglycemic drugs in their combined pharmaceutical dosage form. Spectrochimica ActA PARt A: Molecular and Biomolecular Spectroscopy. 2018; 205: 235-242. http://bit.ly/37207mb
- Lotfy HM, Mohamed D, Mowaka S. A comparative study of smart spectrophotometric methods for simultaneous determination of sitagliptin phosphate and metformin hydrochloride in their binary mixture. Spectrochimica ActA PARt A: Molecular and Biomolecular Spectroscopy. 2015; 149: 441-451. http://bit.ly/38dSw40
- ICH. (2005) Validation of Analytical Procedure: Text and Methodology Q2 (R1). ICH Steering Committee. European Medicines Agency. 1994; 13: 1-15. http://bit.ly/2NBoAad
Sign up for Article Alerts